面瘫(专业版)
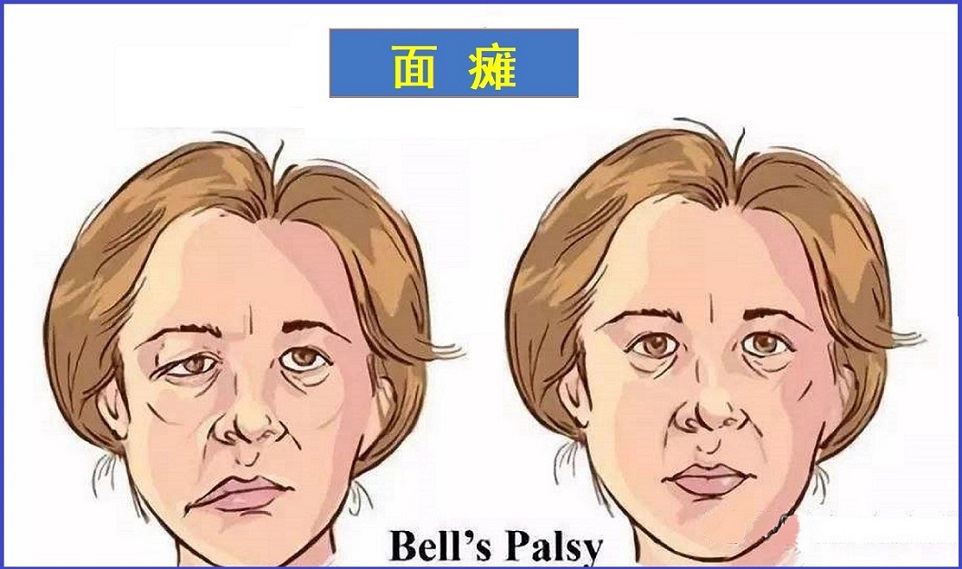
贝尔麻痹是面部一侧突然无力或瘫痪,这种情况一般是暂时性的。
其他名称:贝尔麻痹;面神经麻痹
英文名称:Bell's Palsy
最初的症状可能包括如下:
如果面瘫原因是已知的,则对应治疗,可能包括药物或手术。
综合选项包括如下:
调整饮食与生活方式
营养与草本综合干预
以下是基于循证医学和营养学有关文献综合的结果。
有助于防控面瘫的营养和草本主要包括如下:
促进神经元健康、抑制炎症和降低病毒活性的营养物质可能通过靶向贝尔氏麻痹的潜在机制而带来益处。更多相关内容,可参考本网站专文:慢性炎症管理,以及带状疱疹 >>
1.维生素B12:
维生素B12对神经元健康很重要1,2。在一项对照试验中,对60名持续时间超过两周的贝尔麻痹患者的三种治疗方案进行了比较,第一组每周三次肌肉注射500mcg甲钴胺(活性维生素B12),持续至少八周或直到康复;第二组每天服用60mg泼尼松,持续三周;第三组服用甲钴胺加泼尼松。治疗一周后,甲钴胺和甲钴胺加泼尼松组明显改善,但单独使用泼尼松的组仅略有改善。甲钴胺和甲钴胺加泼尼松组的参与者平均需要大约两周的时间才能完全康复,而仅使用泼尼松的组则需要九周半的时间3。
一项荟萃分析包括了五项随机对照试验,共有344名贝尔麻痹症患者,发现与单独针灸相比,注射维生素B12和针灸可以降低不完全康复的可能性4。由于维生素B12通常是安全且相对便宜的,它是治疗贝尔麻痹症的一种有趣的自然选择5。
2.乙酰L肉碱:
乙酰-L-肉碱是一种遍布中枢和外周神经系统的化合物,在包括脂肪酸加工在内的几个过程中发挥作用。一项针对43名特发性面瘫受试者的随机安慰剂对照试验发现,与甲基强的松龙加安慰剂的受试者相比,每天口服3g乙酰-L-卡碱和50mg甲基强的松,持续14天的受试人面部神经功能恢复更快。经过10天的治疗,乙酰-L-肉碱组的瘫痪程度降低了一半,但安慰剂组没有变化6。
此外,支持使用乙酰-L-肉碱的还有在其他涉及神经损伤的情况下使用该营养素的研究。临床前研究表明,乙酰-L-肉碱可以预防和减轻化疗药物紫杉醇(Abraxane)引起的神经疼痛7。二项临床研究发现,乙酰-L-肉碱对治疗各种形式的神经病变有效,包括糖尿病和药物诱导的神经病变8。
3.其他:
鉴于贝尔麻痹相对良性,其病程通常有限,在大多数情况下预后良好,因此很少有人体临床试验检查营养干预措施。然而,由于贝尔麻痹症的炎症和神经受累,下列促进神经元健康和减轻炎症的营养物质可能会有所帮助。
3.1.烟酸:
烟酸(维生素B3)是一种会导致血管扩张的维生素,这就是为什么它会导致许多人脸红的原因。这种增加的血流量被认为有助于加快面部神经的愈合。一些证据表明,口服或肌肉注射烟酸可能对治疗贝尔麻痹症有用,尽管数据非常有限。
在一组使用100-250mg烟酸治疗的74例较早病例中,除一例外,其余病例在2-4周内均表现出良好的面部神经反应,以至于观察者无法指出哪一侧面部瘫痪。在74例病例中,有39例在瘫痪发作后两天内开始治疗,这些患者在14天内完全康复9。
3.2.欧米伽3脂肪酸:
Omega-3脂肪酸可以减少体内炎症。通常从鱼类中获得的ω-3脂肪酸(EPA和DHA),抑制核因子κB(NF-ĸB)促炎分子的信号传导,并降低引发炎症的其他化学物质的水平,如肿瘤坏死因子-α(TNF-α)和白细胞介素6(IL-6)10。这些脂肪酸也被代谢成一种名为消退素(Resolvins)的化合物,保护神经免受炎症损伤11。
一些临床前证据表明,ω-3脂肪酸可能有助于改善神经损伤后的恢复,而在健康人中,它们已被证明可以改善运动神经功能12。在1型糖尿病患者中,补充鱼油也被证明可以显著改善神经生长13。虽然没有对贝尔麻痹进行任何研究,但有理由认为ω-3脂肪酸可能为炎症性神经损伤和功能恢复提供一些支持。
3.3.α-硫辛酸:
α-硫辛酸是一种参与葡萄糖和脂肪代谢的自由基清除营养素14,15,用于治疗代谢紊乱,如肥胖和糖尿病,以及神经并发症,如糖尿病神经病变、干眼症和视网膜病变16-18。α -硫辛酸已在动物和神经功能障碍的实验室模型以及一些临床研究中被证明可以改善神经功能15。
3.4.银杏叶:
大量研究表明,银杏叶提取物具有神经保护作用19,这可能使其成为治疗贝尔麻痹的有效药物。例如,银杏被发现可以抑制自由基对神经细胞膜的损伤,减少神经炎症,改善神经元线粒体功能和能量产生,并促进神经生长和神经元连接的形成20-23。在临床前研究中,银杏提取物被发现可以促进创伤、毒素暴露、缺血(缺氧)和氧化应激后的神经愈合24-28。在脱髓鞘疾病的动物和细胞模型中,银杏的一种活性成分被发现部分通过减少炎症信号来促进髓鞘的再生21。银杏的一个化合物在实验室中也显示出对疱疹病毒的抗病毒活性29。
3.5.姜黄素:
姜黄素具有强大的抗炎、抗氧化特性,已被证明有助于预防糖尿病和酒精引起的神经损伤,可能是通过保护神经元免受炎症诱导的损伤30,31。具体而言,姜黄素能够通过增加抑制NF-ĸB活性的蛋白质活性来减少炎症,NF-ĸB是炎症的主要触发因素。姜黄素已被证明可以在中风、老年痴呆和氧化应激的临床前模型中保护神经元免受损伤32-35。
3.6.绿茶提取物:
绿茶含有大量被称为儿茶素的多酚,包括表没食子儿茶素没食子酸盐(EGCG),可以清除自由基,抑制氧化损伤,降低氧化应激水平。临床前研究表明,EGCG可以通过抑制神经系统中激活的免疫细胞的炎症信号传导和促进一种重要的神经生长因子脑源性神经营养因子(BDNF)的释放来特异性地保护神经元36,37。
动物研究表明,EGCG还可以减少与代谢紊乱有关的全身和神经系统炎症,而代谢紊乱会增加贝尔氏麻痹的风险38,39。动物模型显示EGCG可能在预防一系列神经系统疾病方面发挥作用36,40,41
3.7.维生素E:
维生素E由生育酚和生育三烯酚组成,具有清除自由基和抗炎作用。由于其作为脂质化合物的性质,它具有特别的神经保护作用,并且已被认为对一系列神经系统疾病有益42-44。对动物的研究表明,维生素E有可能防止氧化和炎症相关的神经损伤44-46。其他动物研究表明,对HSV感染的有效免疫反应需要足够的维生素E水平47。
3.8.甘草:
甘草传统上被用来对抗病毒感染。它含有强烈抗炎作用的化合物,并在临床前研究中显示出对几种病毒的抗病毒活性,包括HSV-1和水痘带状疱疹病毒48-50。
3.9.锌:
锌是正常神经功能的重要营养素,缺乏锌会导致听力和味觉障碍,并导致抑郁和神经疾病51。锌支持神经生长和新的神经元连接的形成52。它还抑制HSV-1的复制,并已在多项临床试验中有效用于缩短疱疹爆发的持续时间53,54。
3.10.赖氨酸:
赖氨酸是有时用于治疗HSV感染的一种氨基酸55。一项双盲、安慰剂对照试验,包括患有口腔面部或生殖器疱疹的参与者,发现他们在六个月内每天三次摄入1g L-赖氨酸,可以降低疱疹爆发的频率、持续时间和严重程度56。其他研究的结果表明,特别是在高于每天3g的剂量下,可以减少HSV引起的唇疱疹的频率、缩短持续时间和降低严重程度,但支持性证据的质量较低57,58。尽管赖氨酸尚未在Bell’s麻痹的背景下进行评估,但其可能的抗疱疹活性表明它可能有帮助。
3.11.灵芝:
灵芝在中国、日本和韩国已经被药用了几个世纪59。研究人员在灵芝中发现了两种不同的化合物,即灵芝蛋白多糖(GLPG)和酸性蛋白结合多糖(APBP),在实验室环境中显示出对HSV-1和HSV-2的强抗病毒活性60-62。灵芝化合物也被证明可以减少炎症、降低氧化应激和增强免疫防御63。
含有灵芝蘑菇的草药制剂在疱疹病毒爆发受试者的非对照临床试验中显示出有希望的结果。据报道,包括灵芝在内的六种草药的组合可以减轻五例带状疱疹引起的疼痛。其他试验报道,同样含有灵芝的草药混合物可以减轻复发性口腔和生殖器疱疹患者的疼痛,缩短症状持续时间64-66。
3.12.香蜂草:
香蜂草传统上用于治疗疱疹爆发和其他病毒感染67。多项实验室实验表明,香蜂草提取物对HSV-1和HSV-2都具有抗病毒活性68-72。临床试验评估了香蜂草外用制剂的疗效,并显示出积极的结果。
在一项有66名参与者参加的双盲、安慰剂对照试验中,香蜂草软膏每天涂抹四次,持续五天,改善了口腔疱疹的症状,缩短了愈合时间,并防止了感染的传播。作者进一步提出,香蜂草可能会增加两次发作之间的时间73。另外两项涉及115和116名患者的试验发现,用香蜂草提取物进行局部治疗可以缓解口腔疱疹症状,并且在早期使用时最为有效74。在实验室和动物模型中,香蜂草也被认为可以保护神经免受炎症、缺氧(低氧水平,如由于血液循环不良)和氧化应激造成的损伤75-78。
3.13.蜂胶:
蜂胶是一种从蜂箱中采集的树脂状物质,具有悠久的药用历史79。它含有多种化合物的混合物,包括黄酮类化合物和其他多酚,具有强烈的抗炎和氧化应激降低作用80。一些蜂胶衍生的化合物已显示出抗微生物活性,包括对HSV-1和HSV-2的抗病毒活性80-82。
全面的综述表明,蜂胶可能对治疗疱疹病毒相关的皮肤病变有益,但现有的研究质量并不高83,84。在嘴唇疱疹病变受试者的临床试验中,0.5%的蜂胶提取物溶液在缩短愈合时间和减轻疼痛、灼热、瘙痒、紧张和肿胀等症状方面比局部使用抗病毒药物阿昔洛韦更有效85,86。蜂胶具有抗病毒和抗炎特性,有可能对贝尔麻痹症有益。
3.14.褐藻糖胶:
褐藻糖胶(Fucoidan)是在可食用褐藻中发现的天然多糖,在临床前模型中被发现可以促进平衡的免疫功能87。218在临床前研究中,它们还可以增强抗病毒免疫反应,并表现出直接的抗病毒作用,包括对抗HSV-1和HSV-288-90。此外,褐藻糖胶具有减少炎症和氧化应激的能力87。一份关于两个病例研究的报告指出,局部使用4%褐藻糖胶霜可显著缩短与复发性口腔疱疹爆发相关的愈合时间91。
3.15.其他抗炎和抗病毒营养素:
其他几种具有抗炎、抗病毒和神经保护特性的营养素可能在加快贝尔麻痹患者的愈合和恢复方面发挥作用。其中包括维生素A、C和D92-97。白藜芦醇和槲皮素等黄酮类化合物,以及富含黄酮类化合物的接骨木提取物,也具有这些特性,有可能帮助促进贝尔氏麻痹的恢复97-99。益生菌可以调节免疫功能,支持神经元健康和再生100。
更多可点击其综合干预方案如下:
以及参阅本网如下专文的相关内容:
医疗干预
常规治疗措施包括如下:
其他疗法
如果面瘫涉及到眼睑,可能需要保护眼睛,包括:
参考文献:
1. Tanaka H. [Old or new medicine? Vitamin B12 and peripheral nerve neuropathy]. Brain Nerve. Sep 2013;65(9):1077-82.
2. Rizzo G, Laganà AS. Chapter 6 - A review of vitamin B12. In: Patel VB, ed. Molecular Nutrition. Academic Press; 2020:105-129.
3. Jalaludin MA. Methylcobalamin treatment of Bell's palsy. Methods and findings in experimental and clinical pharmacology. Oct 1995;17(8):539-44.
4. Wang LL et al. Acupuncture and vitamin B12 injection for Bell's palsy: no high-quality evidence exists. Neural Regen Res. May 2015;10(5):808-13.
5. Sickels M. Treatment options for patients with Bell's palsy. American family physician. Aug 1 2008;78(3):316, 319; author reply 319-20.
6. Mezzina C et al. Idiopathic facial paralysis: new therapeutic prospects with acetyl-L-carnitine. International journal of clinical pharmacology research. 1992;12(5-6):299-304.
7. Flatters SJ et al. Acetyl-L-carnitine prevents and reduces paclitaxel-induced painful peripheral neuropathy. Neurosci Lett. Apr 24 2006;397(3):219-23.
8. Sima AA et al. Acetyl-L-carnitine improves pain, nerve regeneration, and vibratory perception in patients with chronic diabetic neuropathy:an analysis of two randomized placebo-controlled trials. Diabetes Care. Jan 2005;28(1):89-94.
9. Kime CE. Bell's palsy: a new syndrome associated with treatment by nicotinic acid; a guide to adequate medical therapy. AMA Arch Otolaryngol. Jul 1958;68(1):28-32.
10. Calder PC. Omega-3 fatty acids and inflammatory processes: from molecules to man. Biochemical Society transactions. Oct 15 2017;45(5):1105-1115.
11. Chamani S et al. Resolution of Inflammation in Neurodegenerative Diseases: The Role of Resolvins. Mediators Inflamm. 2020;2020:3267172.
12. Gladman SJ et al. Improved outcome after peripheral nerve injury in mice with increased levels of endogenous ω-3 polyunsaturated fatty acids. J Neurosci. Jan 11 2012;32(2):563-71.
13. Lewis EJH et al. Effect of omega-3 supplementation on neuropathy in type 1 diabetes: A 12-month pilot trial. Neurology. Jun 13 2017;88(24):2294-2301.
14. Tutelyan VA et al. [Lipoic acid: physiological role and prospects for clinical application]. Voprosy pitaniia. 2019;88(4):6-11.
15. Salehi B et al. Insights on the Use of α-Lipoic Acid for Therapeutic Purposes. Biomolecules. Aug 9 2019;9(8)
16. Nguyen H, Gupta V. Alpha-Lipoic Acid. StatPearls Publishing Copyright © 2021, StatPearls Publishing LLC.; 2021.
17. Ajith TA. Alpha-lipoic acid: A possible pharmacological agent for treating dry eye disease and retinopathy in diabetes. Clin Exp Pharmacol Physiol. Dec 2020;47(12):1883-1890.
18. Chukanova EI et al. [Alpha-lipoic acid in the treatment of diabetic polyneuropathy]. Zh Nevrol Psikhiatr Im S S Korsakova. 2018;118(1):103-109.
19. Singh SK et al. Neuroprotective and Antioxidant Effect of Ginkgo biloba Extract Against AD and Other Neurological Disorders. Neurotherapeutics: the journal of the American Society for Experimental NeuroTherapeutics . Jul 2019;16(3):666-674.
20. Lejri I et al. Ginkgo biloba extract increases neurite outgrowth and activates the Akt/mTOR pathway. PLoS One. 2019;14(12):e0225761.
21. Yin JJ et al. Dynamic Balance of Microglia and Astrocytes Involved in the Remyelinating Effect of Ginkgolide B. Frontiers in cellular neuroscience. 2019;13:572.
22. Mohammed NA et al. Oral Supplements of <i>Ginkgo biloba</i> Extract Alleviate Neuroinflammation, Oxidative Impairments and Neurotoxicity in Rotenone-Induced Parkinsonian Rats. Current pharmaceutical biotechnology. 2020;21(12):1259-1268.
23. Gargouri B et al. Anti-neuroinflammatory effects of Ginkgo biloba extract EGb761 in LPS-activated primary microglial cells. Phytomedicine. May 15 2018;44:45-55.
24. Al-Adwani DG et al. Neurotherapeutic effects of Ginkgo biloba extract and its terpene trilactone, ginkgolide B, on sciatic crush injury model: A new evidence. PLoS One. 2019;14(12):e0226626.
25. Cho HK et al. Neuroprotective Effect of Ginkgo Biloba Extract Against Hypoxic Retinal Ganglion Cell Degeneration In Vitro and In Vivo. Journal of medicinal food. Aug 2019;22(8):771-778.
26. Kaur S et al. Anti-inflammatory effects of Ginkgo biloba extract against trimethyltin-induced hippocampal neuronal injury. Inflammopharmacol. Feb 2018;26(1):87-104.
27. Zhou X et al. Long-term pre-treatment of antioxidant Ginkgo biloba extract EGb-761 attenuates cerebral-ischemia-induced neuronal damage in aged mice. Biomedicine & pharmacotherapy. Jan 2017;85:256-263.
28. Huang WL et al. Extract of Ginkgo biloba promotes neuronal regeneration in the hippocampus after exposure to acrylamide. Neural Regen Res. Aug 2017;12(8):1287-1293.
29. Borenstein R et al. Ginkgolic acid inhibits fusion of enveloped viruses. Sci Rep. Mar 16 2020;10(1):4746.
30. Kandhare AD et al. Therapeutic role of curcumin in prevention of biochemical and behavioral aberration induced by alcoholic neuropathy in laboratory animals. Neurosci Lett. Mar 5 2012;511(1):18-22.
31. Kulkarni SK et al. An overview of curcumin in neurological disorders. Indian journal of pharmaceutical sciences. Mar 2010;72(2):149-54.
32. Liu ZJ et al. Curcumin Protects Neuron against Cerebral Ischemia-Induced Inflammation through Improving PPAR-Gamma Function. Evidence-based complementary and alternative medicine: eCAM. 2013;2013:470975.
33. Potter PE. Curcumin: a natural substance with potential efficacy in Alzheimer's disease. J Exp Pharmacol. 2013;5:23-31.
34. Scapagnini G et al. Curcumin activates defensive genes and protects neurons against oxidative stress. Antioxid Redox Signal. Mar-Apr 2006;8(3-4):395-403.
35. Huang HC et al. Dual effects of curcumin on neuronal oxidative stress in the presence of Cu(II). Food and chemical toxicology. Jul 2011;49(7):1578-83.
36. Cheng CY et al. Epigallocatechin-3-Gallate-Loaded Liposomes Favor Anti-Inflammation of Microglia Cells and Promote Neuroprotection. International journal of molecular sciences. Mar 16 2021;22(6)
37. Lai SW et al. Regulatory Effects of Neuroinflammatory Responses Through Brain-Derived Neurotrophic Factor Signaling in Microglial Cells. Molecular neurobiology. Sep 2018;55(9):7487-7499.
38. Mao L et al. Green Tea Polyphenol (-)-Epigallocatechin Gallate (EGCG) Attenuates Neuroinflammation in Palmitic Acid-Stimulated BV-2 Microglia and High-Fat Diet-Induced Obese Mice. International journal of molecular sciences. Oct 13 2019;20(20)
39. Zhou J et al. Effects of (-)-Epigallocatechin Gallate (EGCG) on Energy Expenditure and Microglia-Mediated Hypothalamic Inflammation in Mice Fed a High-Fat Diet. Nutrients. Nov 5 2018;10(11)
40. Wang J et al. Protective effect of epigallocatechin-3-gallate against neuroinflammation and anxiety-like behavior in a rat model of myocardial infarction. Brain and behavior. Jun 2020;10(6):e01633.
41. Cheng-Chung Wei J et al. Epigallocatechin gallate attenuates amyloid β-induced inflammation and neurotoxicity in EOC 13.31 microglia. European journal of pharmacology. Jan 5 2016;770:16-24.
42. Tan SW et al. Cellular uptake and anti-inflammatory effects of palm oil-derived delta (δ)-tocotrienol in microglia. Cell Immunol. Nov 2020;357:104200.
43. Manosso LM et al. Vitamin E for the management of major depressive disorder: possible role of the anti-inflammatory and antioxidant systems. Nutritional neuroscience. Dec 14 2020:1-15.
44. Ambrogini P et al. Excitotoxicity, neuroinflammation and oxidant stress as molecular bases of epileptogenesis and epilepsy-derived neurodegeneration: The role of vitamin E. Biochim Biophys Acta Mol Basis Dis. Jun 2019;1865(6):1098-1112.
45. Betti M et al. Dietary supplementation with α-tocopherol reduces neuroinflammation and neuronal degeneration in the rat brain after kainic acid-induced status epilepticus. Free Radic Res. Oct 2011;45(10):1136-42.
46. Ambrogini P et al. Post-seizure α-tocopherol treatment decreases neuroinflammation and neuronal degeneration induced by status epilepticus in rat hippocampus. Molecular neurobiology. Aug 2014;50(1):246-56.
47. Sheridan PA et al. The immune response to herpes simplex virus encephalitis in mice is modulated by dietary vitamin E. J Nutr. Jan 2008;138(1):130-7.
48. Fiore C et al. Antiviral effects of Glycyrrhiza species. Phytother Res. Feb 2008;22(2):141-8. doi:10.1002/ptr.2295
49. Richard SA. Exploring the Pivotal Immunomodulatory and Anti-Inflammatory Potentials of Glycyrrhizic and Glycyrrhetinic Acids. Mediators Inflamm. 2021;2021:6699560.
50. Sun ZG et al. Research Progress of Glycyrrhizic Acid on Antiviral Activity. Mini Rev Med Chem. 2019;19(10):826-832.
51. Kawahara M et al. Zinc, Carnosine, and Neurodegenerative Diseases. Nutrients. Jan 29 2018;10(2)
52. Kumar V et al. Neurobiology of zinc and its role in neurogenesis. European journal of nutrition. Feb 2021;60(1):55-64.
53. Gaby AR. Natural remedies for Herpes simplex. Altern Med Rev. Jun 2006;11(2):93-101.
54. Read SA et al. The Role of Zinc in Antiviral Immunity. Adv Nutr. Jul 1 2019;10(4):696-710.
55. Flodin NW. The metabolic roles, pharmacology, and toxicology of lysine. J Am Coll Nutr. Feb 1997;16(1):7-21.
56. Griffith RS et al. Success of L-lysine therapy in frequently recurrent herpes simplex infection. Treatment and prophylaxis. Dermatologica. 1987;175(4):183-90.
57. Ozden F et al. Clinical success of lysine in association with serumal and salivary presence of HSV-1 in patients with recurrent aphthous ulceration. Journal of experimental and integrative medicine. 2011;1:191-196.
58. Mailoo VJ et al. Lysine for Herpes Simplex Prophylaxis: A Review of the Evidence. Integrative medicine (Encinitas, Calif). Jun 2017;16(3):42-46.
59. Paterson RR. Ganoderma - a therapeutic fungal biofactory. Phytochemistry. Sep 2006;67(18):1985-2001.
60. Kim YS et al. Antiherpetic activities of acidic protein bound polysacchride isolated from Ganoderma lucidum alone and in combinations with interferons. J Ethnopharmacol. Oct 2000;72(3):451-8.
61. Liu J et al. Possible mode of action of antiherpetic activities of a proteoglycan isolated from the mycelia of Ganoderma lucidum in vitro. J Ethnopharmacol. Dec 2004;95(2-3):265-72.
62. Li Z et al. Possible mechanism underlying the antiherpetic activity of a proteoglycan isolated from the mycelia of Ganoderma lucidum in vitro. J Biochem Mol Biol. Jan 31 2005;38(1):34-40.
63. Lu J et al. Molecular mechanisms of bioactive polysaccharides from Ganoderma lucidum (Lingzhi), a review. International journal of biological macromolecules. May 1 2020;150:765-774.
64. Hijikata Y et al. Effect of Ganoderma lucidum on postherpetic neuralgia. The American journal of Chinese medicine. 1998;26(3-4):375-81.
65. Hijikata Y et al. Herbal mixtures containing the mushroom Ganoderma lucidum improve recovery time in patients with herpes genitalis and labialis. J Altern Complement Med. Nov 2007;13(9):985-7.
66. Hijikata Y et al. Effect of an herbal formula containing Ganoderma lucidum on reduction of herpes zoster pain: a pilot clinical trial. Am J Chin Med. 2005;33(4):517-23.
67. Yarnell E, Abascal K, Rountree R. Herbs for Herpes Simplex Infections. Alternative and Complementary Therapies. 04/01 2009;15:69-74.
68. Mazzanti G et al. Inhibitory activity of Melissa officinalis L. extract on Herpes simplex virus type 2 replication. Nat Prod Res. 2008;22(16):1433-40.
69. Astani A et al. Melissa officinalis extract inhibits attachment of herpes simplex virus in vitro. Chemotherapy. 2012;58(1):70-7.
70. Schnitzler P et al. Melissa officinalis oil affects infectivity of enveloped herpesviruses. Phytomedicine: international journal of phytotherapy and phytopharmacology. Sep 2008;15(9):734-40.
71. Allahverdiyev A et al. Antiviral activity of the volatile oils of Melissa officinalis L. against Herpes simplex virus type-2. Phytomedicine. Nov 2004;11(7-8):657-61.
72. Dimitrova Z et al. Antiherpes effect of Melissa officinalis L. extracts. Acta Microbiol Bulg. 1993;29:65-72.
73. Koytchev R et al. Balm mint extract (Lo-701) for topical treatment of recurring herpes labialis. Phytomedicine : international journal of phytotherapy and phytopharmacology. Oct 1999;6(4):225-30.
74. Wölbling RH et al. Local therapy of herpes simplex with dried extract from Melissa officinalis. Phytomedicine : international journal of phytotherapy and phytopharmacology. Jun 1994;1(1):25-31.
75. Bayat M et al. Neuroprotective properties of Melissa officinalis after hypoxic-ischemic injury both in vitro and in vivo. Journal of Faculty of Pharmacy, Tehran University of Medical Sciences. Oct 3 2012;20(1):42.
76. Hosseini SR et al. Coadministration of Dexamethasone and Melissa officinalis Has Neuroprotective Effects in Rat Animal Model with Spinal Cord Injury. Cell J. Apr-Jun 2017;19(1):102-116.
77. López V et aI. Neuroprotective and neurological properties of Melissa officinalis. Neurochem Res. Nov 2009;34(11):1955-61.
78. Soodi M et al. Melissa officinalis Acidic Fraction Protects Cultured Cerebellar Granule Neurons Against Beta Amyloid-Induced Apoptosis and Oxidative Stress. Cell J. Winter 2017;18(4):556-564.
79. U.S. National Library of Medicine. MedlinePlus. Propolis. Updated 3/24/2021. https://medlineplus.gov/druginfo/natural/390.html
80. Almuhayawi MS. Propolis as a novel antibacterial agent. Saudi J Biol Sci. Nov 2020;27(11):3079-3086.
81. Demir S et al. Antiviral activity of different extracts of standardized propolis preparations against HSV. Antiviral therapy. 2020;25(7):353-363.
82. Schnitzler P et al. Antiviral activity and mode of action of propolis extracts and selected compounds. Phytother Res. Jan 2010;24 Suppl 1:S20-8.
83. Münstedt K. Bee products and the treatment of blister-like lesions around the mouth, skin and genitalia caused by herpes viruses-A systematic review. Complement Ther Med. Apr 2019;43:81-84.
84. Sung SH et al. External Use of Propolis for Oral, Skin, and Genital Diseases: A Systematic Review and Meta-Analysis. Evidence-based complementary and alternative medicine: eCAM. 2017;2017:8025752.
85. Jautová J et al. Lip creams with propolis special extract GH 2002 0.5% versus aciclovir 5.0% for herpes labialis (vesicular stage): Randomized, controlled double-blind study. Wien Med Wochenschr. May 2019;169(7-8):193-201.
86. Arenberger P et al. Comparative Study with a Lip Balm Containing 0.5% Propolis Special Extract GH 2002 versus 5% Aciclovir Cream in Patients with Herpes Labialis in the Papular/Erythematous Stage: A Single-blind, Randomized, Two-arm Study. Curr Ther Res Clin Exp. 2018;88:1-7.
87. Luthuli S et al. Therapeutic Effects of Fucoidan: A Review on Recent Studies. Mar Drugs. Aug 21 2019;17(9)
88. Krylova NV et al. The Comparative Analysis of Antiviral Activity of Native and Modified Fucoidans from Brown Algae Fucus evanescens In Vitro and In Vivo. Mar Drugs. Apr 22 2020;18(4)
89. Sinha S et al. Polysaccharides from Sargassum tenerrimum: structural features, chemical modification and anti-viral activity. Phytochemistry. Feb 2010;71(2-3):235-42.
90. Hayashi K et al. Defensive effects of a fucoidan from brown alga Undaria pinnatifida against herpes simplex virus infection. International immunopharmacology. Jan 2008;8(1):109-16.
91. Tsubura S et al. Case report using 4% fucoidan cream for recurrent oral herpes labialis: patient symptoms markedly improved in terms of time to healing and time to loss of discomfort. Dent Open J. 2017;4(2):19-23.
92. Carrera I et al. Neuroprotection with Natural Antioxidants and Nutraceuticals in the Context of Brain Cell Degeneration: The Epigenetic Connection. Current topics in medicinal chemistry. 2019;19(32):2999-3011.
93. Mousavi S et al. Immunomodulatory and Antimicrobial Effects of Vitamin C. Eur J Microbiol Immunol (Bp). Oct 3 2019;9(3):73-79.
94. Teymoori-Rad M et al. The interplay between vitamin D and viral infections. Reviews in medical virology. Mar 2019;29(2):e2032.
95. Farghali M et al. Can Brain Health Be Supported by Vitamin D-Based Supplements? A Critical Review. Brain sciences. Sep 22 2020;10(9)
96. Airavaara M et al. Neurorestoration. Parkinsonism Relat Disord. Jan 2012;18 Suppl 1(0 1):S143-6.
97. Patel S et al. Induction of cellular and molecular immunomodulatory pathways by vitamin A and flavonoids. Expert Opin Biol Ther. 2015;15(10):1411-28.
98. Kumar S et al. Chemistry and biological activities of flavonoids: an overview. The Scientific World Journal. 2013;2013:162750.
99. Spagnuolo C et al. Neuroprotective Role of Natural Polyphenols. Current topics in medicinal chemistry. 2016;16(17):1943-50.
100. Rice MW et al. Gut Microbiota as a Therapeutic Target to Ameliorate the Biochemical, Neuroanatomical, and Behavioral Effects of Traumatic Brain Injuries. Frontiers in neurology. 2019;10:875.
美国梅奥诊所
www.mayoclinic.org
美国疾病防控中心
http://www.cdc.gov
美国国立神经障碍和卒中研究所
http://www.ninds.nih.gov
美国国立公众健康网
www.medlineplus.gov
加拿大公众卫生局
http://www.phac-aspc.gc.ca
免责声明和安全信息
英文名称:Bell's Palsy
定义
贝尔麻痹是面部一侧突然无力或瘫痪,这种情况一般是暂时性的。 控制面部肌肉的神经在通往脸部的路上穿过一条狭窄的骨骼通路,当出现贝尔氏麻痹时,这种神经路径存在发炎、肿胀,这通常与病毒感染有关。除面部肌肉外,神经还会影响眼泪、唾液、味觉和耳朵中间的小骨。病因
贝尔麻痹的确切原因尚不清楚,一般认为是感染或炎症影响神经的结果,包括如下:- 单纯疱疹病毒, 如感冒疮、生殖器疱疹
- 带状疱疹病毒(水痘)
- 流感
- 巨细胞病毒
- 风疹
- 腺病毒(呼吸系统病毒)
- 手足口病毒感染
风险因素
可能导致面瘫的危险因素包括:- 妊娠,尤其在临产前3个月,及产后1周左右
- 自体免疫性疾病
- 糖尿病
- 家庭成员患贝尔麻痹症
- 感染,如普通感冒、流感、莱姆病以及艾滋病等
- 抑制免疫系统的药物或疗法
- 某些药物
- 接触某些毒素
- 吸烟
- 高血压
- 化疗
症状
面瘫症状可能突然出现,在发生几天后消失。最初的症状可能包括如下:
- 耳朵后面疼痛,脸无力和麻痹
- 耳鸣
- 听力轻度障碍
- 在患侧轻微增加对声音的敏感性
- 面部无力或瘫痪,经常在一边
- 患侧虚弱、麻木
- 下垂的嘴角、流口水
- 撕裂
- 无法闭上眼睛,并可导致:
- 干红色的眼睛
- 眼部溃疡
- 感染
- 患侧失去味觉
- 对声音敏感
- 耳痛
- 口齿不清
- 面部肌肉持久收紧·
- 从眼边撕裂
并发症
并发症可能包括如下:- 面神经不可逆转的损伤
- 神经纤维异常再生,导致某些肌肉不自主地收缩
疗法
大多数情况下,不需要治疗。如果症状恶化,或者恢复时间比预期的长,可能需要治疗。如果面瘫原因是已知的,则对应治疗,可能包括药物或手术。
综合选项包括如下:
调整饮食与生活方式
- 健康均衡饮食,富含蔬菜、水果等。
- 低精氨酸或高赖氨酸饮食,可能有助于减缓单纯疱疹病毒的生长并缩短面瘫的持续时间。这些食品包括乳制品、鱼类和鸡肉。
- 不吸烟,或戒烟。
- 经常运动锻炼,提高身体抗病力。
- 学会和运用管理压力。
营养与草本综合干预
以下是基于循证医学和营养学有关文献综合的结果。
有助于防控面瘫的营养和草本主要包括如下:
促进神经元健康、抑制炎症和降低病毒活性的营养物质可能通过靶向贝尔氏麻痹的潜在机制而带来益处。更多相关内容,可参考本网站专文:慢性炎症管理,以及带状疱疹 >>
1.维生素B12:
维生素B12对神经元健康很重要1,2。在一项对照试验中,对60名持续时间超过两周的贝尔麻痹患者的三种治疗方案进行了比较,第一组每周三次肌肉注射500mcg甲钴胺(活性维生素B12),持续至少八周或直到康复;第二组每天服用60mg泼尼松,持续三周;第三组服用甲钴胺加泼尼松。治疗一周后,甲钴胺和甲钴胺加泼尼松组明显改善,但单独使用泼尼松的组仅略有改善。甲钴胺和甲钴胺加泼尼松组的参与者平均需要大约两周的时间才能完全康复,而仅使用泼尼松的组则需要九周半的时间3。
一项荟萃分析包括了五项随机对照试验,共有344名贝尔麻痹症患者,发现与单独针灸相比,注射维生素B12和针灸可以降低不完全康复的可能性4。由于维生素B12通常是安全且相对便宜的,它是治疗贝尔麻痹症的一种有趣的自然选择5。
2.乙酰L肉碱:
乙酰-L-肉碱是一种遍布中枢和外周神经系统的化合物,在包括脂肪酸加工在内的几个过程中发挥作用。一项针对43名特发性面瘫受试者的随机安慰剂对照试验发现,与甲基强的松龙加安慰剂的受试者相比,每天口服3g乙酰-L-卡碱和50mg甲基强的松,持续14天的受试人面部神经功能恢复更快。经过10天的治疗,乙酰-L-肉碱组的瘫痪程度降低了一半,但安慰剂组没有变化6。
此外,支持使用乙酰-L-肉碱的还有在其他涉及神经损伤的情况下使用该营养素的研究。临床前研究表明,乙酰-L-肉碱可以预防和减轻化疗药物紫杉醇(Abraxane)引起的神经疼痛7。二项临床研究发现,乙酰-L-肉碱对治疗各种形式的神经病变有效,包括糖尿病和药物诱导的神经病变8。
3.其他:
鉴于贝尔麻痹相对良性,其病程通常有限,在大多数情况下预后良好,因此很少有人体临床试验检查营养干预措施。然而,由于贝尔麻痹症的炎症和神经受累,下列促进神经元健康和减轻炎症的营养物质可能会有所帮助。
3.1.烟酸:
烟酸(维生素B3)是一种会导致血管扩张的维生素,这就是为什么它会导致许多人脸红的原因。这种增加的血流量被认为有助于加快面部神经的愈合。一些证据表明,口服或肌肉注射烟酸可能对治疗贝尔麻痹症有用,尽管数据非常有限。
在一组使用100-250mg烟酸治疗的74例较早病例中,除一例外,其余病例在2-4周内均表现出良好的面部神经反应,以至于观察者无法指出哪一侧面部瘫痪。在74例病例中,有39例在瘫痪发作后两天内开始治疗,这些患者在14天内完全康复9。
3.2.欧米伽3脂肪酸:
Omega-3脂肪酸可以减少体内炎症。通常从鱼类中获得的ω-3脂肪酸(EPA和DHA),抑制核因子κB(NF-ĸB)促炎分子的信号传导,并降低引发炎症的其他化学物质的水平,如肿瘤坏死因子-α(TNF-α)和白细胞介素6(IL-6)10。这些脂肪酸也被代谢成一种名为消退素(Resolvins)的化合物,保护神经免受炎症损伤11。
一些临床前证据表明,ω-3脂肪酸可能有助于改善神经损伤后的恢复,而在健康人中,它们已被证明可以改善运动神经功能12。在1型糖尿病患者中,补充鱼油也被证明可以显著改善神经生长13。虽然没有对贝尔麻痹进行任何研究,但有理由认为ω-3脂肪酸可能为炎症性神经损伤和功能恢复提供一些支持。
3.3.α-硫辛酸:
α-硫辛酸是一种参与葡萄糖和脂肪代谢的自由基清除营养素14,15,用于治疗代谢紊乱,如肥胖和糖尿病,以及神经并发症,如糖尿病神经病变、干眼症和视网膜病变16-18。α -硫辛酸已在动物和神经功能障碍的实验室模型以及一些临床研究中被证明可以改善神经功能15。
3.4.银杏叶:
大量研究表明,银杏叶提取物具有神经保护作用19,这可能使其成为治疗贝尔麻痹的有效药物。例如,银杏被发现可以抑制自由基对神经细胞膜的损伤,减少神经炎症,改善神经元线粒体功能和能量产生,并促进神经生长和神经元连接的形成20-23。在临床前研究中,银杏提取物被发现可以促进创伤、毒素暴露、缺血(缺氧)和氧化应激后的神经愈合24-28。在脱髓鞘疾病的动物和细胞模型中,银杏的一种活性成分被发现部分通过减少炎症信号来促进髓鞘的再生21。银杏的一个化合物在实验室中也显示出对疱疹病毒的抗病毒活性29。
3.5.姜黄素:
姜黄素具有强大的抗炎、抗氧化特性,已被证明有助于预防糖尿病和酒精引起的神经损伤,可能是通过保护神经元免受炎症诱导的损伤30,31。具体而言,姜黄素能够通过增加抑制NF-ĸB活性的蛋白质活性来减少炎症,NF-ĸB是炎症的主要触发因素。姜黄素已被证明可以在中风、老年痴呆和氧化应激的临床前模型中保护神经元免受损伤32-35。
3.6.绿茶提取物:
绿茶含有大量被称为儿茶素的多酚,包括表没食子儿茶素没食子酸盐(EGCG),可以清除自由基,抑制氧化损伤,降低氧化应激水平。临床前研究表明,EGCG可以通过抑制神经系统中激活的免疫细胞的炎症信号传导和促进一种重要的神经生长因子脑源性神经营养因子(BDNF)的释放来特异性地保护神经元36,37。
动物研究表明,EGCG还可以减少与代谢紊乱有关的全身和神经系统炎症,而代谢紊乱会增加贝尔氏麻痹的风险38,39。动物模型显示EGCG可能在预防一系列神经系统疾病方面发挥作用36,40,41
3.7.维生素E:
维生素E由生育酚和生育三烯酚组成,具有清除自由基和抗炎作用。由于其作为脂质化合物的性质,它具有特别的神经保护作用,并且已被认为对一系列神经系统疾病有益42-44。对动物的研究表明,维生素E有可能防止氧化和炎症相关的神经损伤44-46。其他动物研究表明,对HSV感染的有效免疫反应需要足够的维生素E水平47。
3.8.甘草:
甘草传统上被用来对抗病毒感染。它含有强烈抗炎作用的化合物,并在临床前研究中显示出对几种病毒的抗病毒活性,包括HSV-1和水痘带状疱疹病毒48-50。
3.9.锌:
锌是正常神经功能的重要营养素,缺乏锌会导致听力和味觉障碍,并导致抑郁和神经疾病51。锌支持神经生长和新的神经元连接的形成52。它还抑制HSV-1的复制,并已在多项临床试验中有效用于缩短疱疹爆发的持续时间53,54。
3.10.赖氨酸:
赖氨酸是有时用于治疗HSV感染的一种氨基酸55。一项双盲、安慰剂对照试验,包括患有口腔面部或生殖器疱疹的参与者,发现他们在六个月内每天三次摄入1g L-赖氨酸,可以降低疱疹爆发的频率、持续时间和严重程度56。其他研究的结果表明,特别是在高于每天3g的剂量下,可以减少HSV引起的唇疱疹的频率、缩短持续时间和降低严重程度,但支持性证据的质量较低57,58。尽管赖氨酸尚未在Bell’s麻痹的背景下进行评估,但其可能的抗疱疹活性表明它可能有帮助。
3.11.灵芝:
灵芝在中国、日本和韩国已经被药用了几个世纪59。研究人员在灵芝中发现了两种不同的化合物,即灵芝蛋白多糖(GLPG)和酸性蛋白结合多糖(APBP),在实验室环境中显示出对HSV-1和HSV-2的强抗病毒活性60-62。灵芝化合物也被证明可以减少炎症、降低氧化应激和增强免疫防御63。
含有灵芝蘑菇的草药制剂在疱疹病毒爆发受试者的非对照临床试验中显示出有希望的结果。据报道,包括灵芝在内的六种草药的组合可以减轻五例带状疱疹引起的疼痛。其他试验报道,同样含有灵芝的草药混合物可以减轻复发性口腔和生殖器疱疹患者的疼痛,缩短症状持续时间64-66。
3.12.香蜂草:
香蜂草传统上用于治疗疱疹爆发和其他病毒感染67。多项实验室实验表明,香蜂草提取物对HSV-1和HSV-2都具有抗病毒活性68-72。临床试验评估了香蜂草外用制剂的疗效,并显示出积极的结果。
在一项有66名参与者参加的双盲、安慰剂对照试验中,香蜂草软膏每天涂抹四次,持续五天,改善了口腔疱疹的症状,缩短了愈合时间,并防止了感染的传播。作者进一步提出,香蜂草可能会增加两次发作之间的时间73。另外两项涉及115和116名患者的试验发现,用香蜂草提取物进行局部治疗可以缓解口腔疱疹症状,并且在早期使用时最为有效74。在实验室和动物模型中,香蜂草也被认为可以保护神经免受炎症、缺氧(低氧水平,如由于血液循环不良)和氧化应激造成的损伤75-78。
3.13.蜂胶:
蜂胶是一种从蜂箱中采集的树脂状物质,具有悠久的药用历史79。它含有多种化合物的混合物,包括黄酮类化合物和其他多酚,具有强烈的抗炎和氧化应激降低作用80。一些蜂胶衍生的化合物已显示出抗微生物活性,包括对HSV-1和HSV-2的抗病毒活性80-82。
全面的综述表明,蜂胶可能对治疗疱疹病毒相关的皮肤病变有益,但现有的研究质量并不高83,84。在嘴唇疱疹病变受试者的临床试验中,0.5%的蜂胶提取物溶液在缩短愈合时间和减轻疼痛、灼热、瘙痒、紧张和肿胀等症状方面比局部使用抗病毒药物阿昔洛韦更有效85,86。蜂胶具有抗病毒和抗炎特性,有可能对贝尔麻痹症有益。
3.14.褐藻糖胶:
褐藻糖胶(Fucoidan)是在可食用褐藻中发现的天然多糖,在临床前模型中被发现可以促进平衡的免疫功能87。218在临床前研究中,它们还可以增强抗病毒免疫反应,并表现出直接的抗病毒作用,包括对抗HSV-1和HSV-288-90。此外,褐藻糖胶具有减少炎症和氧化应激的能力87。一份关于两个病例研究的报告指出,局部使用4%褐藻糖胶霜可显著缩短与复发性口腔疱疹爆发相关的愈合时间91。
3.15.其他抗炎和抗病毒营养素:
其他几种具有抗炎、抗病毒和神经保护特性的营养素可能在加快贝尔麻痹患者的愈合和恢复方面发挥作用。其中包括维生素A、C和D92-97。白藜芦醇和槲皮素等黄酮类化合物,以及富含黄酮类化合物的接骨木提取物,也具有这些特性,有可能帮助促进贝尔氏麻痹的恢复97-99。益生菌可以调节免疫功能,支持神经元健康和再生100。
更多可点击其综合干预方案如下:
以及参阅本网如下专文的相关内容:
医疗干预
常规治疗措施包括如下:
- 皮质类固醇药:可以减轻炎症。以及可能使用抗病毒药物结合皮质类固醇。不过,单独使用抗病毒药物没有疗效。
- 人造眼泪和眼科软膏可用于防止角膜损伤。
- 手术治疗:在某些情况下,手术可以减轻面神经的压迫。
其他疗法
如果面瘫涉及到眼睑,可能需要保护眼睛,包括:
- 物理治疗以改善面瘫的一些功能
- 心理咨询:可以帮助患者管理情绪问题,因为面瘫症状可能令人苦恼。
- 润肤剂或滴眼液
- 包扎眼睛
- 按摩面部肌肉也可能有所帮助。
预防
仍然没有预防贝尔麻痹症的指南。参考文献:
1. Tanaka H. [Old or new medicine? Vitamin B12 and peripheral nerve neuropathy]. Brain Nerve. Sep 2013;65(9):1077-82.
2. Rizzo G, Laganà AS. Chapter 6 - A review of vitamin B12. In: Patel VB, ed. Molecular Nutrition. Academic Press; 2020:105-129.
3. Jalaludin MA. Methylcobalamin treatment of Bell's palsy. Methods and findings in experimental and clinical pharmacology. Oct 1995;17(8):539-44.
4. Wang LL et al. Acupuncture and vitamin B12 injection for Bell's palsy: no high-quality evidence exists. Neural Regen Res. May 2015;10(5):808-13.
5. Sickels M. Treatment options for patients with Bell's palsy. American family physician. Aug 1 2008;78(3):316, 319; author reply 319-20.
6. Mezzina C et al. Idiopathic facial paralysis: new therapeutic prospects with acetyl-L-carnitine. International journal of clinical pharmacology research. 1992;12(5-6):299-304.
7. Flatters SJ et al. Acetyl-L-carnitine prevents and reduces paclitaxel-induced painful peripheral neuropathy. Neurosci Lett. Apr 24 2006;397(3):219-23.
8. Sima AA et al. Acetyl-L-carnitine improves pain, nerve regeneration, and vibratory perception in patients with chronic diabetic neuropathy:an analysis of two randomized placebo-controlled trials. Diabetes Care. Jan 2005;28(1):89-94.
9. Kime CE. Bell's palsy: a new syndrome associated with treatment by nicotinic acid; a guide to adequate medical therapy. AMA Arch Otolaryngol. Jul 1958;68(1):28-32.
10. Calder PC. Omega-3 fatty acids and inflammatory processes: from molecules to man. Biochemical Society transactions. Oct 15 2017;45(5):1105-1115.
11. Chamani S et al. Resolution of Inflammation in Neurodegenerative Diseases: The Role of Resolvins. Mediators Inflamm. 2020;2020:3267172.
12. Gladman SJ et al. Improved outcome after peripheral nerve injury in mice with increased levels of endogenous ω-3 polyunsaturated fatty acids. J Neurosci. Jan 11 2012;32(2):563-71.
13. Lewis EJH et al. Effect of omega-3 supplementation on neuropathy in type 1 diabetes: A 12-month pilot trial. Neurology. Jun 13 2017;88(24):2294-2301.
14. Tutelyan VA et al. [Lipoic acid: physiological role and prospects for clinical application]. Voprosy pitaniia. 2019;88(4):6-11.
15. Salehi B et al. Insights on the Use of α-Lipoic Acid for Therapeutic Purposes. Biomolecules. Aug 9 2019;9(8)
16. Nguyen H, Gupta V. Alpha-Lipoic Acid. StatPearls Publishing Copyright © 2021, StatPearls Publishing LLC.; 2021.
17. Ajith TA. Alpha-lipoic acid: A possible pharmacological agent for treating dry eye disease and retinopathy in diabetes. Clin Exp Pharmacol Physiol. Dec 2020;47(12):1883-1890.
18. Chukanova EI et al. [Alpha-lipoic acid in the treatment of diabetic polyneuropathy]. Zh Nevrol Psikhiatr Im S S Korsakova. 2018;118(1):103-109.
19. Singh SK et al. Neuroprotective and Antioxidant Effect of Ginkgo biloba Extract Against AD and Other Neurological Disorders. Neurotherapeutics: the journal of the American Society for Experimental NeuroTherapeutics . Jul 2019;16(3):666-674.
20. Lejri I et al. Ginkgo biloba extract increases neurite outgrowth and activates the Akt/mTOR pathway. PLoS One. 2019;14(12):e0225761.
21. Yin JJ et al. Dynamic Balance of Microglia and Astrocytes Involved in the Remyelinating Effect of Ginkgolide B. Frontiers in cellular neuroscience. 2019;13:572.
22. Mohammed NA et al. Oral Supplements of <i>Ginkgo biloba</i> Extract Alleviate Neuroinflammation, Oxidative Impairments and Neurotoxicity in Rotenone-Induced Parkinsonian Rats. Current pharmaceutical biotechnology. 2020;21(12):1259-1268.
23. Gargouri B et al. Anti-neuroinflammatory effects of Ginkgo biloba extract EGb761 in LPS-activated primary microglial cells. Phytomedicine. May 15 2018;44:45-55.
24. Al-Adwani DG et al. Neurotherapeutic effects of Ginkgo biloba extract and its terpene trilactone, ginkgolide B, on sciatic crush injury model: A new evidence. PLoS One. 2019;14(12):e0226626.
25. Cho HK et al. Neuroprotective Effect of Ginkgo Biloba Extract Against Hypoxic Retinal Ganglion Cell Degeneration In Vitro and In Vivo. Journal of medicinal food. Aug 2019;22(8):771-778.
26. Kaur S et al. Anti-inflammatory effects of Ginkgo biloba extract against trimethyltin-induced hippocampal neuronal injury. Inflammopharmacol. Feb 2018;26(1):87-104.
27. Zhou X et al. Long-term pre-treatment of antioxidant Ginkgo biloba extract EGb-761 attenuates cerebral-ischemia-induced neuronal damage in aged mice. Biomedicine & pharmacotherapy. Jan 2017;85:256-263.
28. Huang WL et al. Extract of Ginkgo biloba promotes neuronal regeneration in the hippocampus after exposure to acrylamide. Neural Regen Res. Aug 2017;12(8):1287-1293.
29. Borenstein R et al. Ginkgolic acid inhibits fusion of enveloped viruses. Sci Rep. Mar 16 2020;10(1):4746.
30. Kandhare AD et al. Therapeutic role of curcumin in prevention of biochemical and behavioral aberration induced by alcoholic neuropathy in laboratory animals. Neurosci Lett. Mar 5 2012;511(1):18-22.
31. Kulkarni SK et al. An overview of curcumin in neurological disorders. Indian journal of pharmaceutical sciences. Mar 2010;72(2):149-54.
32. Liu ZJ et al. Curcumin Protects Neuron against Cerebral Ischemia-Induced Inflammation through Improving PPAR-Gamma Function. Evidence-based complementary and alternative medicine: eCAM. 2013;2013:470975.
33. Potter PE. Curcumin: a natural substance with potential efficacy in Alzheimer's disease. J Exp Pharmacol. 2013;5:23-31.
34. Scapagnini G et al. Curcumin activates defensive genes and protects neurons against oxidative stress. Antioxid Redox Signal. Mar-Apr 2006;8(3-4):395-403.
35. Huang HC et al. Dual effects of curcumin on neuronal oxidative stress in the presence of Cu(II). Food and chemical toxicology. Jul 2011;49(7):1578-83.
36. Cheng CY et al. Epigallocatechin-3-Gallate-Loaded Liposomes Favor Anti-Inflammation of Microglia Cells and Promote Neuroprotection. International journal of molecular sciences. Mar 16 2021;22(6)
37. Lai SW et al. Regulatory Effects of Neuroinflammatory Responses Through Brain-Derived Neurotrophic Factor Signaling in Microglial Cells. Molecular neurobiology. Sep 2018;55(9):7487-7499.
38. Mao L et al. Green Tea Polyphenol (-)-Epigallocatechin Gallate (EGCG) Attenuates Neuroinflammation in Palmitic Acid-Stimulated BV-2 Microglia and High-Fat Diet-Induced Obese Mice. International journal of molecular sciences. Oct 13 2019;20(20)
39. Zhou J et al. Effects of (-)-Epigallocatechin Gallate (EGCG) on Energy Expenditure and Microglia-Mediated Hypothalamic Inflammation in Mice Fed a High-Fat Diet. Nutrients. Nov 5 2018;10(11)
40. Wang J et al. Protective effect of epigallocatechin-3-gallate against neuroinflammation and anxiety-like behavior in a rat model of myocardial infarction. Brain and behavior. Jun 2020;10(6):e01633.
41. Cheng-Chung Wei J et al. Epigallocatechin gallate attenuates amyloid β-induced inflammation and neurotoxicity in EOC 13.31 microglia. European journal of pharmacology. Jan 5 2016;770:16-24.
42. Tan SW et al. Cellular uptake and anti-inflammatory effects of palm oil-derived delta (δ)-tocotrienol in microglia. Cell Immunol. Nov 2020;357:104200.
43. Manosso LM et al. Vitamin E for the management of major depressive disorder: possible role of the anti-inflammatory and antioxidant systems. Nutritional neuroscience. Dec 14 2020:1-15.
44. Ambrogini P et al. Excitotoxicity, neuroinflammation and oxidant stress as molecular bases of epileptogenesis and epilepsy-derived neurodegeneration: The role of vitamin E. Biochim Biophys Acta Mol Basis Dis. Jun 2019;1865(6):1098-1112.
45. Betti M et al. Dietary supplementation with α-tocopherol reduces neuroinflammation and neuronal degeneration in the rat brain after kainic acid-induced status epilepticus. Free Radic Res. Oct 2011;45(10):1136-42.
46. Ambrogini P et al. Post-seizure α-tocopherol treatment decreases neuroinflammation and neuronal degeneration induced by status epilepticus in rat hippocampus. Molecular neurobiology. Aug 2014;50(1):246-56.
47. Sheridan PA et al. The immune response to herpes simplex virus encephalitis in mice is modulated by dietary vitamin E. J Nutr. Jan 2008;138(1):130-7.
48. Fiore C et al. Antiviral effects of Glycyrrhiza species. Phytother Res. Feb 2008;22(2):141-8. doi:10.1002/ptr.2295
49. Richard SA. Exploring the Pivotal Immunomodulatory and Anti-Inflammatory Potentials of Glycyrrhizic and Glycyrrhetinic Acids. Mediators Inflamm. 2021;2021:6699560.
50. Sun ZG et al. Research Progress of Glycyrrhizic Acid on Antiviral Activity. Mini Rev Med Chem. 2019;19(10):826-832.
51. Kawahara M et al. Zinc, Carnosine, and Neurodegenerative Diseases. Nutrients. Jan 29 2018;10(2)
52. Kumar V et al. Neurobiology of zinc and its role in neurogenesis. European journal of nutrition. Feb 2021;60(1):55-64.
53. Gaby AR. Natural remedies for Herpes simplex. Altern Med Rev. Jun 2006;11(2):93-101.
54. Read SA et al. The Role of Zinc in Antiviral Immunity. Adv Nutr. Jul 1 2019;10(4):696-710.
55. Flodin NW. The metabolic roles, pharmacology, and toxicology of lysine. J Am Coll Nutr. Feb 1997;16(1):7-21.
56. Griffith RS et al. Success of L-lysine therapy in frequently recurrent herpes simplex infection. Treatment and prophylaxis. Dermatologica. 1987;175(4):183-90.
57. Ozden F et al. Clinical success of lysine in association with serumal and salivary presence of HSV-1 in patients with recurrent aphthous ulceration. Journal of experimental and integrative medicine. 2011;1:191-196.
58. Mailoo VJ et al. Lysine for Herpes Simplex Prophylaxis: A Review of the Evidence. Integrative medicine (Encinitas, Calif). Jun 2017;16(3):42-46.
59. Paterson RR. Ganoderma - a therapeutic fungal biofactory. Phytochemistry. Sep 2006;67(18):1985-2001.
60. Kim YS et al. Antiherpetic activities of acidic protein bound polysacchride isolated from Ganoderma lucidum alone and in combinations with interferons. J Ethnopharmacol. Oct 2000;72(3):451-8.
61. Liu J et al. Possible mode of action of antiherpetic activities of a proteoglycan isolated from the mycelia of Ganoderma lucidum in vitro. J Ethnopharmacol. Dec 2004;95(2-3):265-72.
62. Li Z et al. Possible mechanism underlying the antiherpetic activity of a proteoglycan isolated from the mycelia of Ganoderma lucidum in vitro. J Biochem Mol Biol. Jan 31 2005;38(1):34-40.
63. Lu J et al. Molecular mechanisms of bioactive polysaccharides from Ganoderma lucidum (Lingzhi), a review. International journal of biological macromolecules. May 1 2020;150:765-774.
64. Hijikata Y et al. Effect of Ganoderma lucidum on postherpetic neuralgia. The American journal of Chinese medicine. 1998;26(3-4):375-81.
65. Hijikata Y et al. Herbal mixtures containing the mushroom Ganoderma lucidum improve recovery time in patients with herpes genitalis and labialis. J Altern Complement Med. Nov 2007;13(9):985-7.
66. Hijikata Y et al. Effect of an herbal formula containing Ganoderma lucidum on reduction of herpes zoster pain: a pilot clinical trial. Am J Chin Med. 2005;33(4):517-23.
67. Yarnell E, Abascal K, Rountree R. Herbs for Herpes Simplex Infections. Alternative and Complementary Therapies. 04/01 2009;15:69-74.
68. Mazzanti G et al. Inhibitory activity of Melissa officinalis L. extract on Herpes simplex virus type 2 replication. Nat Prod Res. 2008;22(16):1433-40.
69. Astani A et al. Melissa officinalis extract inhibits attachment of herpes simplex virus in vitro. Chemotherapy. 2012;58(1):70-7.
70. Schnitzler P et al. Melissa officinalis oil affects infectivity of enveloped herpesviruses. Phytomedicine: international journal of phytotherapy and phytopharmacology. Sep 2008;15(9):734-40.
71. Allahverdiyev A et al. Antiviral activity of the volatile oils of Melissa officinalis L. against Herpes simplex virus type-2. Phytomedicine. Nov 2004;11(7-8):657-61.
72. Dimitrova Z et al. Antiherpes effect of Melissa officinalis L. extracts. Acta Microbiol Bulg. 1993;29:65-72.
73. Koytchev R et al. Balm mint extract (Lo-701) for topical treatment of recurring herpes labialis. Phytomedicine : international journal of phytotherapy and phytopharmacology. Oct 1999;6(4):225-30.
74. Wölbling RH et al. Local therapy of herpes simplex with dried extract from Melissa officinalis. Phytomedicine : international journal of phytotherapy and phytopharmacology. Jun 1994;1(1):25-31.
75. Bayat M et al. Neuroprotective properties of Melissa officinalis after hypoxic-ischemic injury both in vitro and in vivo. Journal of Faculty of Pharmacy, Tehran University of Medical Sciences. Oct 3 2012;20(1):42.
76. Hosseini SR et al. Coadministration of Dexamethasone and Melissa officinalis Has Neuroprotective Effects in Rat Animal Model with Spinal Cord Injury. Cell J. Apr-Jun 2017;19(1):102-116.
77. López V et aI. Neuroprotective and neurological properties of Melissa officinalis. Neurochem Res. Nov 2009;34(11):1955-61.
78. Soodi M et al. Melissa officinalis Acidic Fraction Protects Cultured Cerebellar Granule Neurons Against Beta Amyloid-Induced Apoptosis and Oxidative Stress. Cell J. Winter 2017;18(4):556-564.
79. U.S. National Library of Medicine. MedlinePlus. Propolis. Updated 3/24/2021. https://medlineplus.gov/druginfo/natural/390.html
80. Almuhayawi MS. Propolis as a novel antibacterial agent. Saudi J Biol Sci. Nov 2020;27(11):3079-3086.
81. Demir S et al. Antiviral activity of different extracts of standardized propolis preparations against HSV. Antiviral therapy. 2020;25(7):353-363.
82. Schnitzler P et al. Antiviral activity and mode of action of propolis extracts and selected compounds. Phytother Res. Jan 2010;24 Suppl 1:S20-8.
83. Münstedt K. Bee products and the treatment of blister-like lesions around the mouth, skin and genitalia caused by herpes viruses-A systematic review. Complement Ther Med. Apr 2019;43:81-84.
84. Sung SH et al. External Use of Propolis for Oral, Skin, and Genital Diseases: A Systematic Review and Meta-Analysis. Evidence-based complementary and alternative medicine: eCAM. 2017;2017:8025752.
85. Jautová J et al. Lip creams with propolis special extract GH 2002 0.5% versus aciclovir 5.0% for herpes labialis (vesicular stage): Randomized, controlled double-blind study. Wien Med Wochenschr. May 2019;169(7-8):193-201.
86. Arenberger P et al. Comparative Study with a Lip Balm Containing 0.5% Propolis Special Extract GH 2002 versus 5% Aciclovir Cream in Patients with Herpes Labialis in the Papular/Erythematous Stage: A Single-blind, Randomized, Two-arm Study. Curr Ther Res Clin Exp. 2018;88:1-7.
87. Luthuli S et al. Therapeutic Effects of Fucoidan: A Review on Recent Studies. Mar Drugs. Aug 21 2019;17(9)
88. Krylova NV et al. The Comparative Analysis of Antiviral Activity of Native and Modified Fucoidans from Brown Algae Fucus evanescens In Vitro and In Vivo. Mar Drugs. Apr 22 2020;18(4)
89. Sinha S et al. Polysaccharides from Sargassum tenerrimum: structural features, chemical modification and anti-viral activity. Phytochemistry. Feb 2010;71(2-3):235-42.
90. Hayashi K et al. Defensive effects of a fucoidan from brown alga Undaria pinnatifida against herpes simplex virus infection. International immunopharmacology. Jan 2008;8(1):109-16.
91. Tsubura S et al. Case report using 4% fucoidan cream for recurrent oral herpes labialis: patient symptoms markedly improved in terms of time to healing and time to loss of discomfort. Dent Open J. 2017;4(2):19-23.
92. Carrera I et al. Neuroprotection with Natural Antioxidants and Nutraceuticals in the Context of Brain Cell Degeneration: The Epigenetic Connection. Current topics in medicinal chemistry. 2019;19(32):2999-3011.
93. Mousavi S et al. Immunomodulatory and Antimicrobial Effects of Vitamin C. Eur J Microbiol Immunol (Bp). Oct 3 2019;9(3):73-79.
94. Teymoori-Rad M et al. The interplay between vitamin D and viral infections. Reviews in medical virology. Mar 2019;29(2):e2032.
95. Farghali M et al. Can Brain Health Be Supported by Vitamin D-Based Supplements? A Critical Review. Brain sciences. Sep 22 2020;10(9)
96. Airavaara M et al. Neurorestoration. Parkinsonism Relat Disord. Jan 2012;18 Suppl 1(0 1):S143-6.
97. Patel S et al. Induction of cellular and molecular immunomodulatory pathways by vitamin A and flavonoids. Expert Opin Biol Ther. 2015;15(10):1411-28.
98. Kumar S et al. Chemistry and biological activities of flavonoids: an overview. The Scientific World Journal. 2013;2013:162750.
99. Spagnuolo C et al. Neuroprotective Role of Natural Polyphenols. Current topics in medicinal chemistry. 2016;16(17):1943-50.
100. Rice MW et al. Gut Microbiota as a Therapeutic Target to Ameliorate the Biochemical, Neuroanatomical, and Behavioral Effects of Traumatic Brain Injuries. Frontiers in neurology. 2019;10:875.
参考来源:
美国梅奥诊所
www.mayoclinic.org
美国疾病防控中心
http://www.cdc.gov
美国国立神经障碍和卒中研究所
http://www.ninds.nih.gov
美国国立公众健康网
www.medlineplus.gov
加拿大公众卫生局
http://www.phac-aspc.gc.ca
免责声明和安全信息
- 本信息(包括任何附带资料)不是为了取代医生或有关合格从业人士的建议或忠告。
- 任何人如果想要对本文涉及的药物、饮食、运动或其他生活方式的使用、或改变调整,以预防或治疗某一特定健康状况或疾病,应首先咨询医生或有关合格从业人士,并获得他/她们的许可。妊娠和哺乳妇女在使用本网站任何内容前,尤其应征求医生的意见。
- 除非另有说明,本网站所述内容仅适用于成人。
- 本网站所推荐的任何产品,消费者应该以实际的产品标签内容为准,尤其应关注重要的安全信息以及产品最新信息,包括剂量、使用方法和禁忌症等。
- 由于循证医学研究、文献及有关产品处于不断的变化中,本网站工作人员将尽力更新。
- 本网站不能保证所载文章内容、综合干预方案以及相关成分或产品述及的健康益处,也不承担任何责任。

